ANALYTICAL SPECTROSCOPY
by Raymond P. W. Scott
D.Sc., F.R.S.C., C.Chem., C.Sci. F.A.I.C, F.C.S.
Essential Information for the Analytical Chemist
Specialising in custom-designed, precision scientific instruments, built, programmed and calibrated
to the most exacting standards. The range includes precision dataloging barographs,
with built-in statistical analysis, Barographic Transient Event Recorders
and computer-interfaced detectors and sensors
for environmental monitoring & process control.

A site dedicated to scientific techniques, experimental methods, &
investigative tools for the inventor, researcher
and laboratory pioneer. Articles on glassblowing, electronics, metalcasting, magnetic
measurements with new material added continually. Check it out!
www.drkfs.net
Practical Aspects of UV/vis Absorption Spectroscopy
A
large molecule will exhibit a range of adsorption lines in it’s
UV Spectrum arising from both atomic
and molecular
orbitals. These lines will result from the different energy absorbing
sources in the atoms, and combination of atoms, that will be unique
to the structure of the compound. As already reported, (2πr)
must be an integral number of wavelengths, i.e.,(n)
Consequently, the absorption bands are frequently so close that they
often cannot be observed individually and merely give a broad band in
the UV adsorption Spectrum as shown for ethyl ethanoate in figure 4A.
However, aromatic substances and unsaturated substances are example
of substances that do give spectra with some fine structure and thus,
can be used by the analyst for structure elucidation or compound
identification (see figure 4B). All the same, most substances will
absorb light in the UV range and, thus, although their spectra are
mostly inadequate for analytical purposes, UV absorption can be used
very effectively for detection in liquid chromatography.
Before
discussing spectroscopic equipment, the use of UV absorption for the
quantitative determination of substances in solution must be
considered. Consequentially, the relationship between solute
concentration and UV absorption has to be examined. The relationship
between the energy of the light transmitted thorough a cell
containing a solution of a UV absorbing substance was examined by
Beer.
Beers law for
Light Absorption
The
relationship between the intensity of light transmitted through a
cell (IT)
and the concentration of solute in the cell, (c),
is given by Beer's Law.
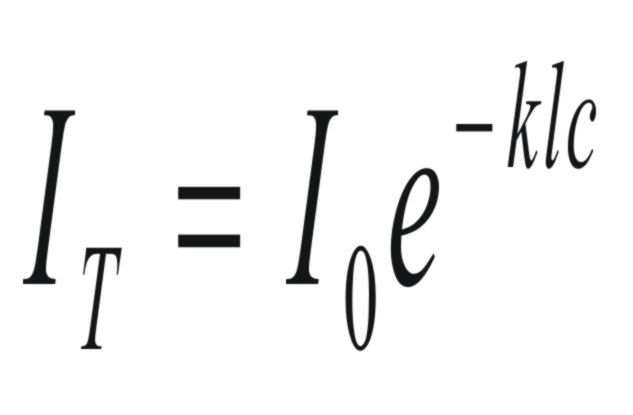 (3)
(3)
or
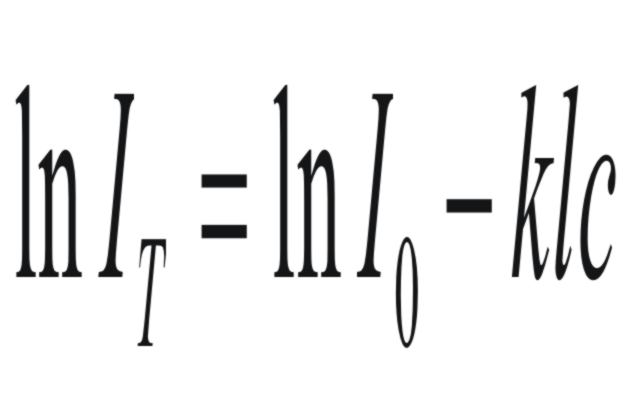
|
where
(Io)
|
is
the intensity of the light entering the cell,
|
|
(l)
|
is
the path length of the cell,
|
|
and
(k)
|
is
the molar extinction coefficient of the solute for the specific
wavelength of the UV Light.
|
If equation (3)
is put in the form,
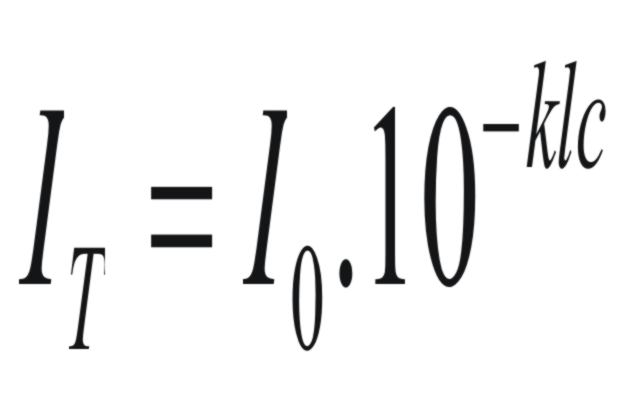 (4)
(4)
then
(K)
is termed the molar extinction coefficient.
Differentiating
equation (4), 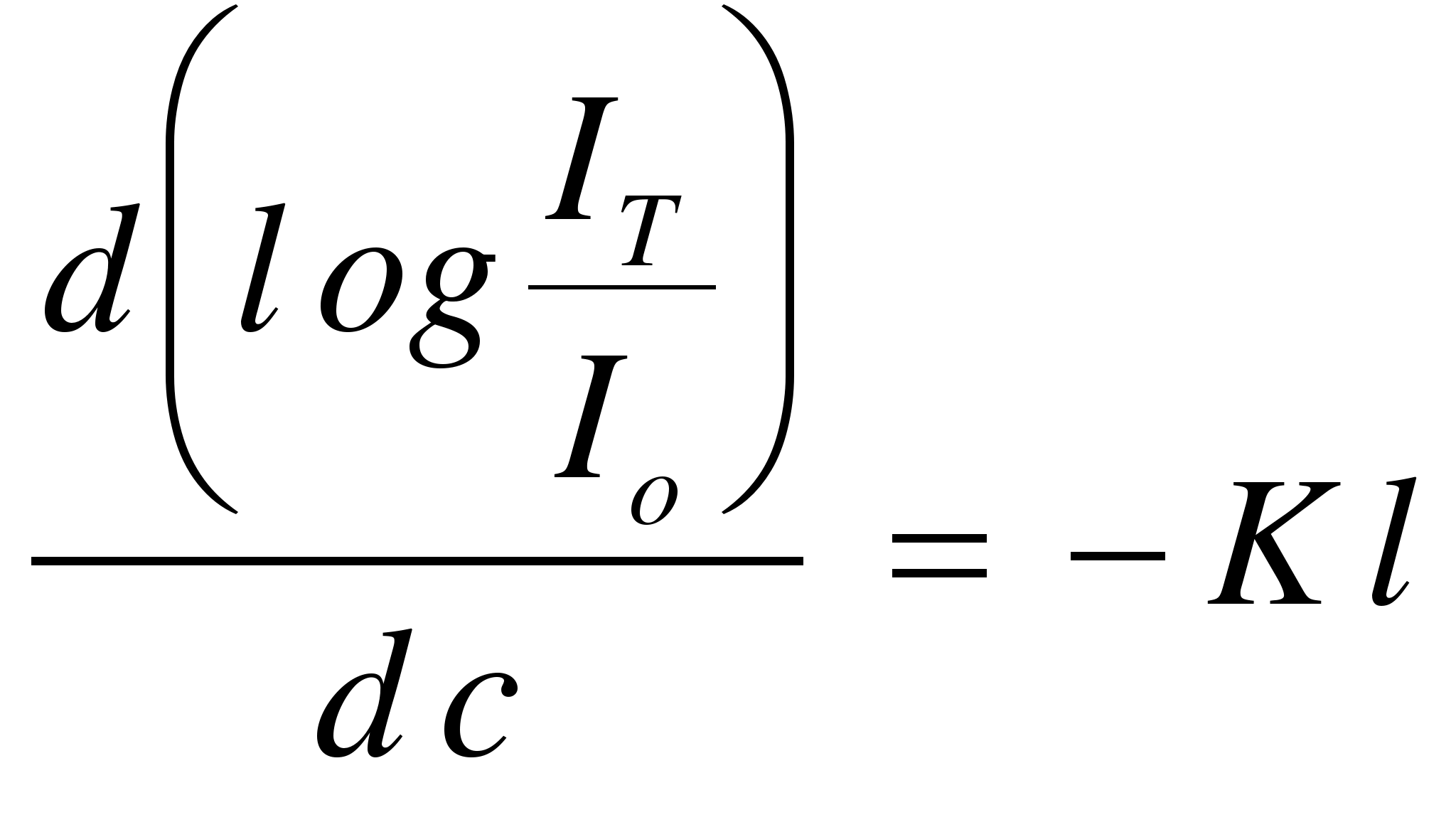 (5)
(5)
It
is seen that the sensitivity of the detector, as measured by the
intensity of the transmitted light will be directly proportional to
the value of the extinction coefficient (K)
and the path length of the cell (l).
Most modern UV detector sensors have path lengths that range between
5 and 20 mm and internal radii that range from about 1 to 10 mm. The
UV spectrometer is a very sensitive instrument and, under suitable
circumstances, spectra can be obtained from concentrations as low as
10-8
g/ml of sample. Measurements being made from a cell 8 mm long and 1
mm in diameter this would correspond to a mass of 0.63 ng
From
equation (5),
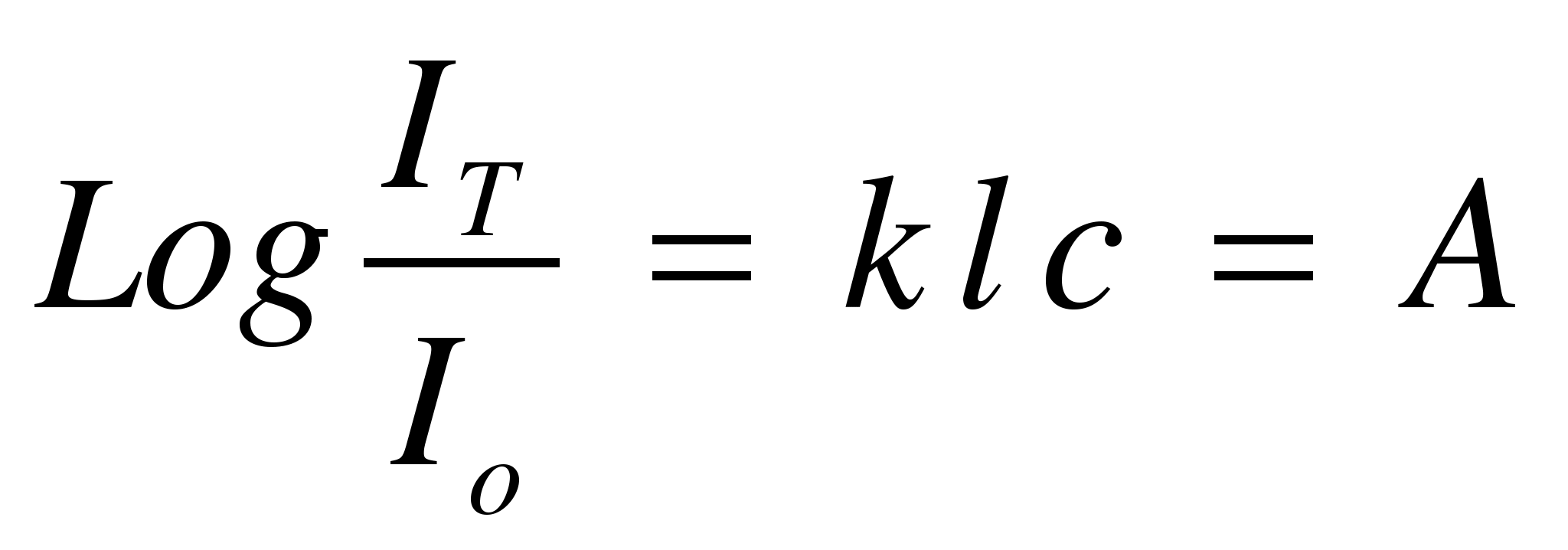 (6)
(6)
where
(A)
is termed the absorbance.
There are a
number of situations where there can be serious deviations from Beers
law. Such deviations usually arise from interactions between the
solute itself or the solute and solvent in which it is dissolved. An
example would be the light absorption by alcohol in an inert solvent
such as isooctane . At low concentrations the alcohol molecules would
exist as single molecules. As the concentration of alcohol was
increased the molecules of alcohol would begin to associate with one
another and the concentration of the monomer would be smaller than it
should be. In some cases the solutes can interact with the solvent
with the same effect. However, if solutions are kept sufficiently
dilute and the solvent is carefully chosen not to interact with the
solute, the deviation from Beers law can be maintained minimal.

About the Author
RAYMOND PETER WILLIAM SCOTT was born on June 20 1924 in Erith, Kent, UK. He studied at the
University of London, obtaining his B.Sc. degree in 1946 and his D.Sc. degree in 1960.
After spending more than a decade at Benzole Producers, Ltd. Where he became head of
the Physical Chemistry Laboratory, he moved to Unilever Research Laboratories as
Manager of their Physical Chemistry department. In 1969 he became Director of Physical
Chemistry at Hoffmann-La Roche, Nutley, NJ, U.S.A. and subsequently accepted the position
of Director of the Applied Research Department at the Perkin-Elmer Corporation, Norwalk, CT, U.S.A.
In 1986 he became an independent consultant and was appointed Visiting Professor at Georgetown
University, Washington, DC, U.S.A. and at Berkbeck College of the University of London; in 1986
he retired but continues to write technical books dealing with various aspects of physical chemistry
and physical chemical techniques. Dr. Scott has authored or co-authored over 200 peer reviewed
scientific papers and authored, co-authored or edited over thirty books on various aspects of
physical and analytical chemistry. Dr. Scott was a founding member of the British chromatography
Society and received the American Chemical society Award in chromatography (1977), the
M. S. Tswett chromatography Medal (1978), the Tswett chromatography Medal U.S.S.R., (1979),
the A. J. P. Martin chromatography Award (1982) and the Royal Society of Chemistry Award in
Analysis and Instrumentation (1988).
Dr. Scott’s activities in gas chromatography started at the inception of the technique,
inventing the Heat of Combustion Detector (the precursor of the Flame Ionization Detector),
pioneered work on high sensitivity detectors, high efficiency columns and presented fundamental
treatments of the relationship between the theory and practice of the technique.
He established the viability of the moving bed continuous preparative gas chromatography,
examined both theoretically and experimentally those factors that controlled dispersion
in packed beds and helped establish the gas chromatograph as a process monitoring instrument.
Dr. Scott took and active part in the renaissance of liquid chromatography,
was involved in the development of high performance liquid chromatography and invented
the wire transport detector. He invented the liquid chromatography mass spectrometry
transport interface, introduced micro-bore liquid chromatography columns and used them
to provide columns of 750,000 theoretical plates and liquid chromatography separations
in less than a second.
Dr. Scott has always been a “hands-on” scientist with a remarkable record of accomplishments in chromatography ranging from hardware design to the development of fundamental theory. He has never shied away from questioning “conventional wisdom” and his original approach to problems has often produced significant breakthroughs.


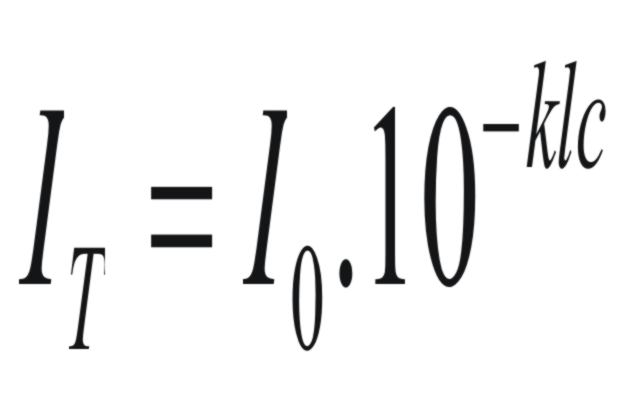 (4)
(4)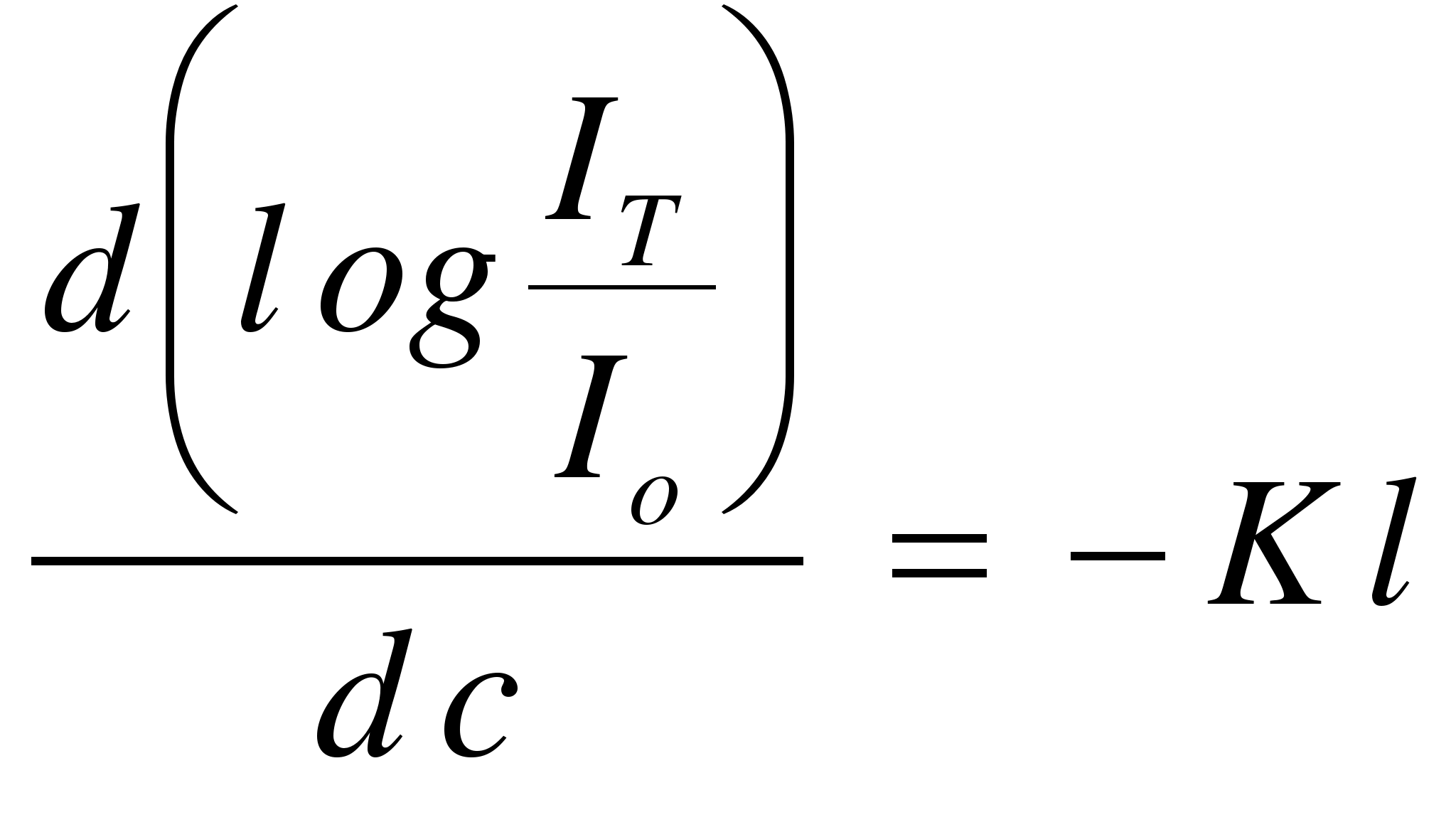 (5)
(5)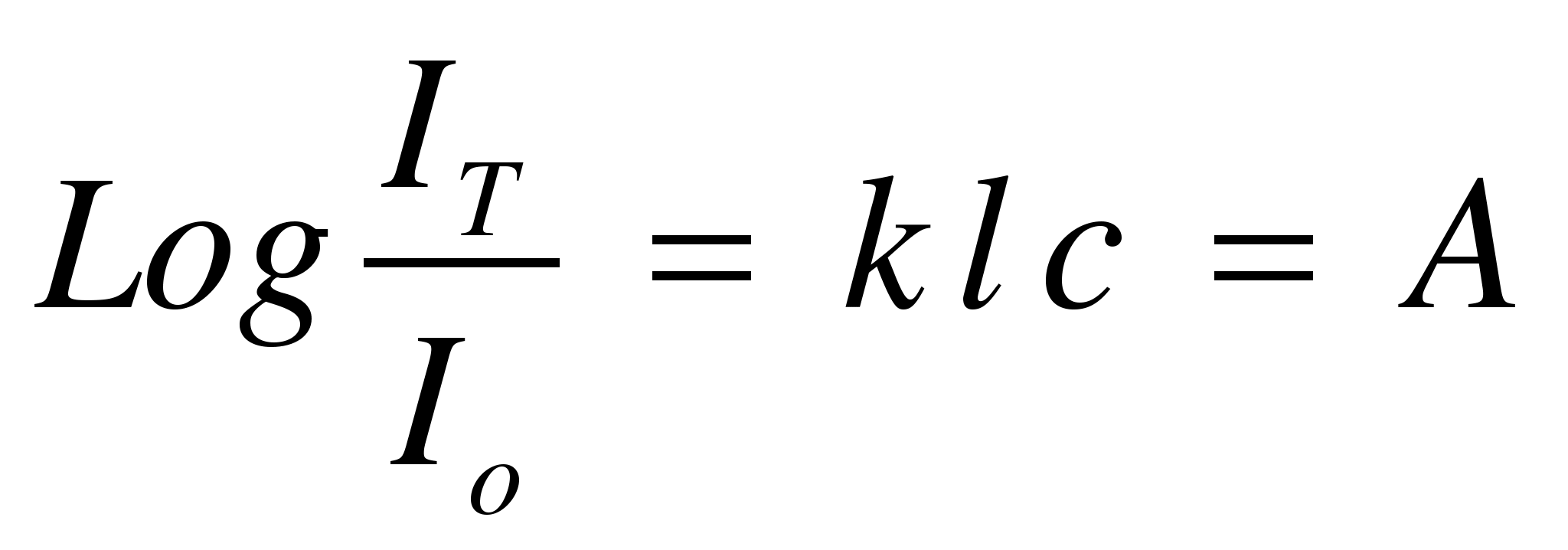 (6)
(6)