ANALYTICAL SPECTROSCOPY
by Raymond P. W. Scott
D.Sc., F.R.S.C., C.Chem., C.Sci. F.A.I.C, F.C.S.
Essential Information for the Analytical Chemist
Specialising in custom-designed, precision scientific instruments, built, programmed and calibrated
to the most exacting standards. The range includes precision dataloging barographs,
with built-in statistical analysis, Barographic Transient Event Recorders
and computer-interfaced detectors and sensors
for environmental monitoring & process control.

A site dedicated to scientific techniques, experimental methods, &
investigative tools for the inventor, researcher
and laboratory pioneer. Articles on glassblowing, electronics, metalcasting, magnetic
measurements with new material added continually. Check it out!
www.drkfs.net
The NMR Spectrometer
Due to the limited techniques
available in the early days of NMR, conventional iron cored
electromagnets or ceramic permanent magnets had to be used to produce
very strong magnetic fields and so field strengths were limited to
about 14 kilogauss. As a result, the early NMR spectrometers could
only operate at about 60 MHz. Today, very strong magnetic fields can
be obtained by employing Superconducting Magnets. It is seen
from Table 1, that for proton spectroscopy, magnetic fields of about
60 and 180 kilogauss respectively will allow the use of frequencies
of 259 or even 750 Mhz and so, modern high-resolution NMR
spectrometers operate at these frequencies. Although hardly used
today, in order to illustrate the relative simplicity of the NMR
instrument, the original low resolution 60 MHz permanent magnet NMR
spectrometer will be briefly described.
A diagram of a 60 MHz permanent
magnet NMR spectrometer is shown in figure 5.

The Perkin Elmer Corporation
pioneered the production of the permanent magnet, 60MHz, NMR machine
in the early 1960s and a simple diagrammatic form of the instrument
is shown in figure 5. This instrument includes the basic parts of all
NMR spectrometers and although modern instruments have more
sophisticated adjustments and electronics and employ superconducting
magnets they all have the basic parts of the original 60 MHz
instrument of Perkin Elmer. The first requirement is a magnet having
the dimensions and strength required for the radiation frequency to
be used. In the above instrument this consisted of a ceramic
permanent magnet having a field-strength of about 1400 gauss. The
second requirement is a coil to provide a small magnetic field that
is adjusted to scan the range appropriate for the protons to be
examined. This coil can vary in complexity and in the original
instrument includes a series of coils (called the Golay coils
(after Marcel Golay the inventor of the capillary column in gas
chromatography)) that could also be used adjust the field to a
maximum degree of homogeneity. The thirds requirement is an RF
coil that both supplies the radiation to the sample and also senses
the energy absorption at the positions of resonance. Finally there is
a tube to contain the sample that is attached to an air turbine to
rotate the sample at high speeds. The rotation of the sample is
essential to ensure that the sample to
be exposed to a magnetic field that is as homogeneous
as possible. As the sample rotates, the net field experienced is the
average of that swept out during a single rotation. The effect of
spinning the sample on resolution is shown in figure 6.
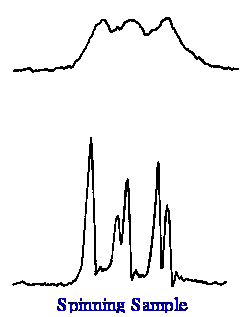
It is seen that the effect of
spinning the sample is quite dramatic and is essential as it would be
impossible to obtain high-resolution spectra without using the sample
spinning technique. Field homogeneity of better than one part in 109
is necessary to obtain a resolution of 1 Hz in a field of 176 500Mhz.

About the Author
RAYMOND PETER WILLIAM SCOTT was born on June 20 1924 in Erith, Kent, UK. He studied at the
University of London, obtaining his B.Sc. degree in 1946 and his D.Sc. degree in 1960.
After spending more than a decade at Benzole Producers, Ltd. Where he became head of
the Physical Chemistry Laboratory, he moved to Unilever Research Laboratories as
Manager of their Physical Chemistry department. In 1969 he became Director of Physical
Chemistry at Hoffmann-La Roche, Nutley, NJ, U.S.A. and subsequently accepted the position
of Director of the Applied Research Department at the Perkin-Elmer Corporation, Norwalk, CT, U.S.A.
In 1986 he became an independent consultant and was appointed Visiting Professor at Georgetown
University, Washington, DC, U.S.A. and at Berkbeck College of the University of London; in 1986
he retired but continues to write technical books dealing with various aspects of physical chemistry
and physical chemical techniques. Dr. Scott has authored or co-authored over 200 peer reviewed
scientific papers and authored, co-authored or edited over thirty books on various aspects of
physical and analytical chemistry. Dr. Scott was a founding member of the British chromatography
Society and received the American Chemical society Award in chromatography (1977), the
M. S. Tswett chromatography Medal (1978), the Tswett chromatography Medal U.S.S.R., (1979),
the A. J. P. Martin chromatography Award (1982) and the Royal Society of Chemistry Award in
Analysis and Instrumentation (1988).
Dr. Scott’s activities in gas chromatography started at the inception of the technique,
inventing the Heat of Combustion Detector (the precursor of the Flame Ionization Detector),
pioneered work on high sensitivity detectors, high efficiency columns and presented fundamental
treatments of the relationship between the theory and practice of the technique.
He established the viability of the moving bed continuous preparative gas chromatography,
examined both theoretically and experimentally those factors that controlled dispersion
in packed beds and helped establish the gas chromatograph as a process monitoring instrument.
Dr. Scott took and active part in the renaissance of liquid chromatography,
was involved in the development of high performance liquid chromatography and invented
the wire transport detector. He invented the liquid chromatography mass spectrometry
transport interface, introduced micro-bore liquid chromatography columns and used them
to provide columns of 750,000 theoretical plates and liquid chromatography separations
in less than a second.
Dr. Scott has always been a “hands-on” scientist with a remarkable record of accomplishments in chromatography ranging from hardware design to the development of fundamental theory. He has never shied away from questioning “conventional wisdom” and his original approach to problems has often produced significant breakthroughs.




