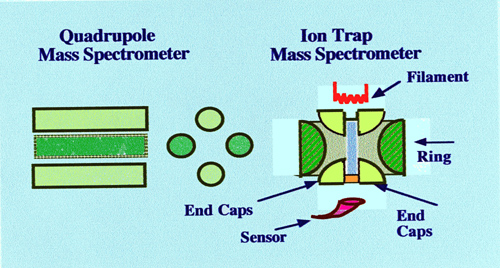
The Ion Trap Detector
The
ion trap detector is a modified form of
the Quadrupole Mass Spectrometer, but was originally designed more
specifically as a chromatography detector than as a instrument for
structure elucidation or identification. Nevertheless the combination
of the Ion Trap Mass Spectrometer with the chromatograph is another
powerful tandem technique. The electrode orientation of the
quadrupole Ion Trap Mass Spectrometer is shown in figure 76.
It
was shown in figure 68 that the
quadrupole spectrometer contains four, rod electrodes. The ion trap
mass spectrometer has a quite different electrode arrangement and
consists of three cylindrically symmetrical electrodes comprised of
two end caps and a ring. The device can be made very small the
opposite internal electrode faces being only 2 cm apart. Each
electrode has accurately machined hyperbolic internal faces.
In
a similar manner to the quadrupole
spectrometer, an rf voltage together with an
additional DC
voltage is applied to the ring and the end caps are grounded. In the
same way as the Quadrupole Mass Spectrometer, the rf
voltage
causes rapid reversals of field direction, so any ions are
alternately accelerated and decelerated in the axial direction and
vice versa in the radial direction. Again the
operating
parameters, (a), and (q),
define the conditions of oscillation which are analogous to those for
the Quadrupole Mass Spectrometer but, in this case, (r0)
is the internal radius of the ring electrode.
The
ion trap is small and (ro)
is typically about 1 cm. At a chosen voltage, ions of a specific mass
range are held oscillating in the trap. Initially, the electron beam
is used to produce ions and after a given time the beam is turned
off. All the ions, except those selected by the magnitude of the
applied rf voltage, are lost to the walls of the
trap, and the
remainder continue oscillating in the trap. The potential of the
applied rf voltage is then increased, and the ions
sequentially assume unstable trajectories and leave the trap via the
aperture to the sensor. The ions exit the trap in order of their
increasing m/z values.
The
first Ion Trap Mass Spectrometers were
not very efficient, but it was found that the introduction traces of
helium to the ion trap significantly improved the quality of the
spectra. The improvement appeared to result from ion–helium
collisions that reduced the energy of the ions and allow them to
concentrate in the center of the trap. The spectra produced are quite
satisfactory for solute identification by comparison with reference
spectra. However, the Spectrum produced for a given substance will
probably differ considerably from that produced by the normal
Quadrupole Mass Spectrometer.
A
commercial Ion Trap Mass Spectrometer,
Varian 500 MS mass spectrometer is shown in figure 77.
The
device incorporates a new enhanced
charge capacity system that extends the number of ions that can be
stored in resonation which, in turn, increases the over all
signal-to-noise ratio of the system. The spectrometer is fitted with
an electro-spray ionization and atmospheric pressure ionization
facilities, the latter being temperature programmable and, thus,
improves the ionization efficiency of thermally labile materials. The
two ionization systems can be exchanged in less than a minute.

Courtesy of Varian Inc.
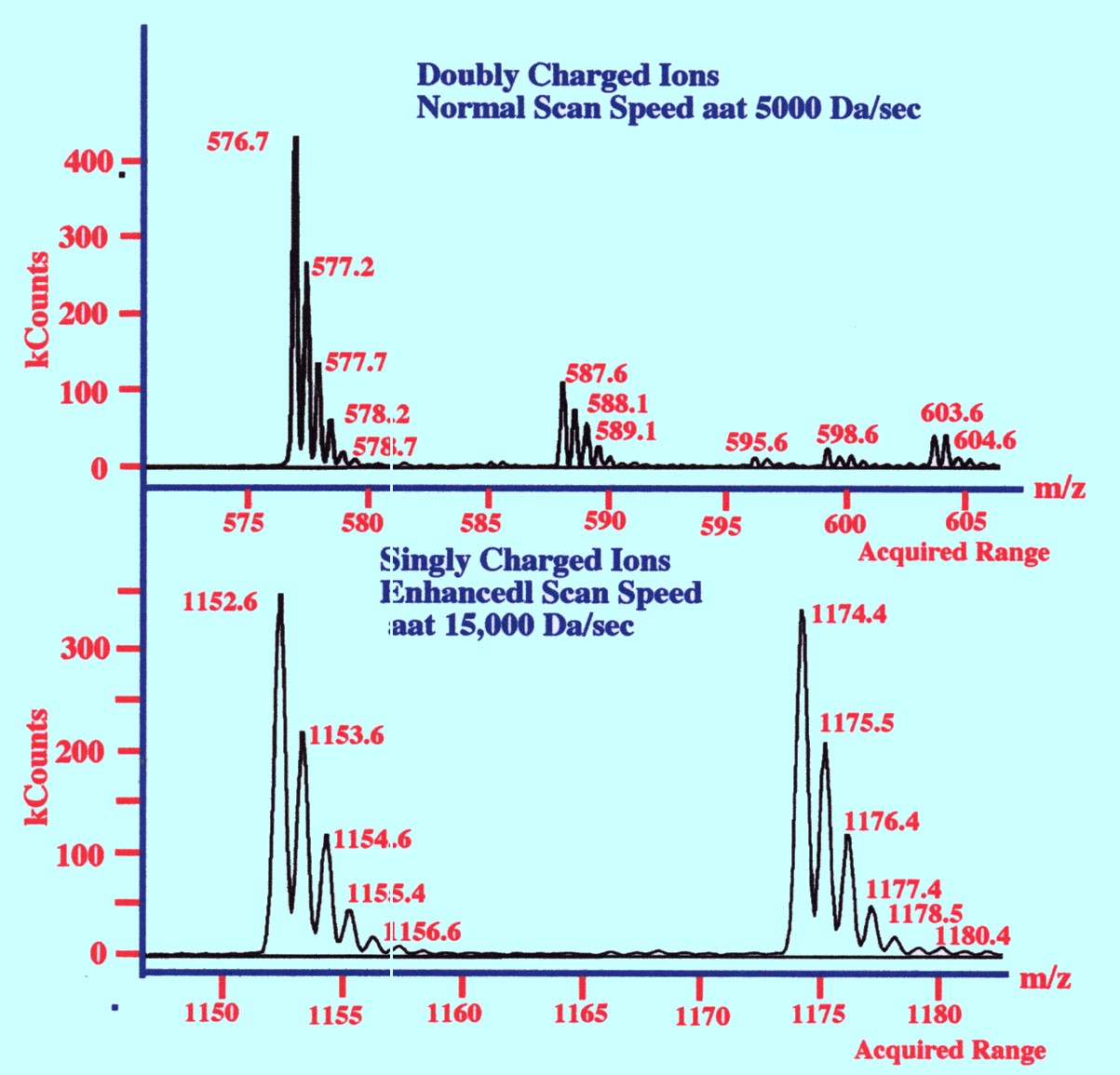
The
‘electro spray’-‘ion
trap’ system readily produces multiple charged ions and as the
mass analyzer discriminates on the basis of (m/z),
((m) is mass and (z)
is the charge) and not just mass, then a doubly charged ion of mass
1000 will appear on the m/z scale at 500. Thus, multiple charged ions
in effect extends the mass range of the mass spectrometer.
The
heights of peaks having multiple
charges are linearly relate to one another and, thus, by fitting a
linear function to the data the presence of multiple charge ions can
be exposed. An example of multiple charges is shown by the spectra in
figure 78.
Courtesy of Varian Inc.
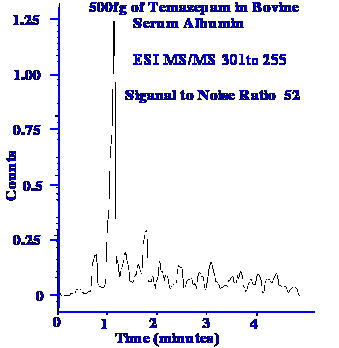
The
Ion Trap Mass Spectrometer is extremely sensitive as shown by the
chromatogram that was monitored by the spectrometer given in figure
79. It is seen that the peak represents a sample size of 500 fg. It
should also be noted that the signal-to-noise ratio is 52. Now,
assuming the peak is discernable when its height is twice the noise,
then the ultimate sensitivity will be about ca.
19 fg.

About the Author
RAYMOND PETER WILLIAM SCOTT was born on June 20 1924 in Erith, Kent, UK. He studied at the
University of London, obtaining his B.Sc. degree in 1946 and his D.Sc. degree in 1960.
After spending more than a decade at Benzole Producers, Ltd. Where he became head of
the Physical Chemistry Laboratory, he moved to Unilever Research Laboratories as
Manager of their Physical Chemistry department. In 1969 he became Director of Physical
Chemistry at Hoffmann-La Roche, Nutley, NJ, U.S.A. and subsequently accepted the position
of Director of the Applied Research Department at the Perkin-Elmer Corporation, Norwalk, CT, U.S.A.
In 1986 he became an independent consultant and was appointed Visiting Professor at Georgetown
University, Washington, DC, U.S.A. and at Berkbeck College of the University of London; in 1986
he retired but continues to write technical books dealing with various aspects of physical chemistry
and physical chemical techniques. Dr. Scott has authored or co-authored over 200 peer reviewed
scientific papers and authored, co-authored or edited over thirty books on various aspects of
physical and analytical chemistry. Dr. Scott was a founding member of the British chromatography
Society and received the American Chemical society Award in chromatography (1977), the
M. S. Tswett chromatography Medal (1978), the Tswett chromatography Medal U.S.S.R., (1979),
the A. J. P. Martin chromatography Award (1982) and the Royal Society of Chemistry Award in
Analysis and Instrumentation (1988).
Dr. Scott’s activities in gas chromatography started at the inception of the technique,
inventing the Heat of Combustion Detector (the precursor of the Flame Ionization Detector),
pioneered work on high sensitivity detectors, high efficiency columns and presented fundamental
treatments of the relationship between the theory and practice of the technique.
He established the viability of the moving bed continuous preparative gas chromatography,
examined both theoretically and experimentally those factors that controlled dispersion
in packed beds and helped establish the gas chromatograph as a process monitoring instrument.
Dr. Scott took and active part in the renaissance of liquid chromatography,
was involved in the development of high performance liquid chromatography and invented
the wire transport detector. He invented the liquid chromatography mass spectrometry
transport interface, introduced micro-bore liquid chromatography columns and used them
to provide columns of 750,000 theoretical plates and liquid chromatography separations
in less than a second.
Dr. Scott has always been a “hands-on” scientist with a remarkable record of accomplishments in chromatography ranging from hardware design to the development of fundamental theory. He has never shied away from questioning “conventional wisdom” and his original approach to problems has often produced significant breakthroughs.